 
ArQule’s current discovery efforts are focused on the identification and development of novel kinase inhibitors, leveraging the Company’s proprietary library of compounds. ArQule’s pipeline includes: ARQ 531, an orally bioavailable, potent and reversible inhibitor of both wild type and C481S-mutant BTK, in Phase 1 for patients with B-cell malignancies refractory to other therapeutic options Miransertib (ARQ 092), a selective inhibitor of the AKT serine/threonine kinase, in a Phase 1/2 company-sponsored study for Overgrowth Diseases, in a Phase 1 study for ultra-rare Proteus syndrome conducted by the National Institutes of Health (NIH), and in Phase 1b in combination with the hormonal therapy, anastrozole, in patients with advanced endometrial cancer ARQ 751, a next generation AKT inhibitor, in Phase 1 for patients with AKT1 and PI3K mutations Derazantinib, a multi-kinase inhibitor designed to preferentially inhibit the fibroblast growth factor receptor (FGFR) family, in a registrational trial for iCCA and ARQ 761, a β-lapachone analog being evaluated as a promoter of NQO1-mediated programmed cancer cell necrosis, in Phase 1/2 in multiple oncology indications in partnership with the University of Texas Southwestern Medical Center. Our clinical-stage pipeline consists of five drug candidates, all of which are in targeted, biomarker-defined patient populations, making ArQule a leader among companies our size in precision medicine. ArQule’s mission is to discover, develop and commercialize novel small molecule drugs in areas of high unmet need that will dramatically extend and improve the lives of our patients. We’d like to thank our academic collaborators and the patients and their families for their support and tremendous dedication to this cause.”ĪrQule is a biopharmaceutical company engaged in the research and development of targeted therapeutics to treat cancers and rare diseases. “The reduction in CCTN lesions were particularly striking since these lesions are, in general, relentlessly progressive, and cause severe morbidity, ulcerations and intractable pain. 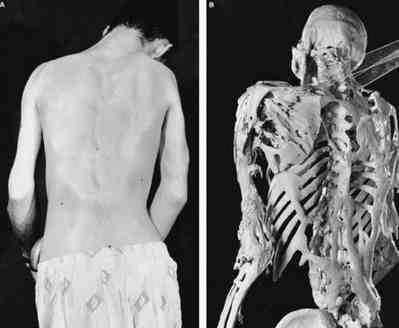
Brian Schwartz, Chief Medical Officer at ArQule. “These data are highly encouraging and support further investigation into the potential use of miransertib for this devastating condition,” said Dr. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
